Research with HeLa Cells
Being a vicious cancer cell in its own right, the HeLa cell line was originally derived for use in cancer research. HeLa cells are extremely malignant, even when compared to other cancer cells from malignant sources. Telomere shortening, which is implicated in eventual cell death after a number of divisions, is prevented in HeLa by the enzyme telomerase which restores telomeres during mitosis. This is why HeLa cells are considered immortal and do not experience cell death after a set number of cell divisions like normal cells other cancer cells.
Even though HeLa cells are very hardy, the HeLa cell line is fairly easy to transfect and very receptive to viral vectors, making it a popular research tool in virology. Since their initiation, HeLa cells have been used in transfection experiments all over the world to study viruses. It was the HeLa cell line that took up the polio virus and made the vaccine that eventually eradicated polio. HeLa is also receptive to Human adenovirus 3, Encephalomyocarditis virus and Human polioviruses 1, 2, and 3.
HeLa Cell Transfection Protocol
A pre-optimized HeLa Transfection Kit is available from Altogen Biosystems, which includes:
- HeLa Transfection Reagent (0.5 ml / 1.5 ml / 8.0 ml)
- Transfection Enhancer (0.5 ml), and
- Complex Condenser (0.5 ml)
The kit is optimized to transfect miRNA, siRNA or DNA plasmid following either forward or reverse transfection protocols. Below is the optimized protocol to transfect a 24-well plate of HeLa cells:
- Plate 7,500- 12,000 HeLa cells per well in 0.5 ml of complete growth medium 12-24 hours prior to transfection
- Wash with 1xPBS and add 0.5 ml of fresh growth medium
- Prepare transfection complexes by mixing 40 µl of serum-free medium, 5.5 µl of transfection reagent, and (Referred to a final volume including growth medium)
- 750 ng DNA (or mRNA), or
- 30 nM – 50 nM of siRNA (or microRNA)
- Incubate transfection complexes at RT for 15-30 minutes
- Optional:Add 2 µl of Complex Condenser. This reagent increases transfection efficiency by reducing the size of transfection complex; however, it may increase cell toxicity
- Add prepared transfection complexes to 0.5 ml of complete growth medium with HeLa cells (from step 2)
- Incubate cells at 37ºC in a humidified CO2incubator
- Assay for phenotype or target gene expression 48-72 hours after transfection
Optional: Adding Transfection Enhancer reagent can increase transfection efficiency. Add 2 µl of Transfection Enhancer reagent 12-24 hours after transfection
If the viability of HeLa cells being transfected is affected at 16-24 hours post-transfection, changing the growth medium and eliminating redundant exposure of cells to transfectant can reduce the level of cytotoxicity
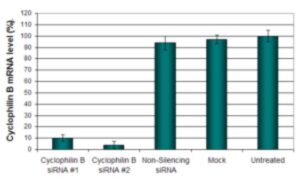
Figure. HeLa cells were transfected with CyclophilinB siRNA and quantified by qRT-PCR following the recommended transfection protocol. CyclophilinB mRNA expression levels were measured 48 hours post-transfection. 18S rRNA levels were used as a housekeeping gene to normalize the CyclophilinB data. Values are normalized to the non-treated sample and data are presented as means ± SD (n=3).
Transfection Controls
Three controls should be included on every transfection plate: a negative control, positive control and non-treated control.
Positive Controls
A positive control ensures the system is working and delivery conditions are optimal. Results of the positive control helps in understanding any day to day experimental differences. Positive controls consists of these characteristics:
- Gene is a housekeeping gene: Ensures target gene is abundantly expressed in all cells and at constant levels
- Abundant expression: Enables easy quantitation of gene expression mRNA or protein levels
- Non-lethal: Altering expression levels of the positive control should not affect phenotype or viability
- Common targets: CyclophilinB (PPIB), Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) and Lamin (LMNA)
Negative Controls
Negative controls have no known derived target and are commonly called a “scrambled” control. The ideal negative control mimics the test compound in G/C content, length and contains identical base modifications. Negative control samples are analyzed the same as test article samples. Negative controls exhibit these characteristics:
- Gene targets: Alien sequences are used (no known target) so that it does not alter expression levels of mRNA or protein
- Non-lethal: Does not affect viability or global gene expression from non-target design
Non-Treated Controls
Non-treated and mock transfection controls (i.e. transfection reagent only) are also essential. The mock and negative controls help to understand experimental toxicity when expression data is compared to the non-treated samples.
Links
Purchase HeLa Transfection Reagent
List of Companies Offering Lab Services
Transfection. Cells and Molecular Biology Research Methods, Protocols, and Lab Techniques
HeLa Cell Line │ Cell Culture Information │ Transfec Information │Get HeLa Transfection Reagenttion